Are chemistry Regents hard?Ĭhemistry is hard. Every year, the Algebra 1 Regents exam is administered in three months: August, January, and June. What happens if you fail a Regents exam? Please do not worry if you fail your Regents Algebra 1 exam! You will typically get another chance to retake the exam. Under the proposal, students would be able to substitute a second Regents Exam in math or science or a vocational exam for this requirement. Global History and Geography is the most frequently failed examination. By working through different problems, you will grow familiar with the way the test is structured and build your knowledge of earth science criteria. One of the best ways to prepare for the NYS Earth Science Regents Exam is by doing practice tests and quizzes while you study. How do I study for Earth Science Regents? How do you find an element on the periodic table? A multiple-choice question offers four possible answers from which you choose the one that best answers the question or completes the statement. This exam contains 85 questions in total. The Chemistry Regents is divided into three parts that test-takers will have three hours to complete. How many questions are on the chemistry regents? Metals at the bottom are least easily oxidized and Nonmetals at the bottom are least easily reduced. 
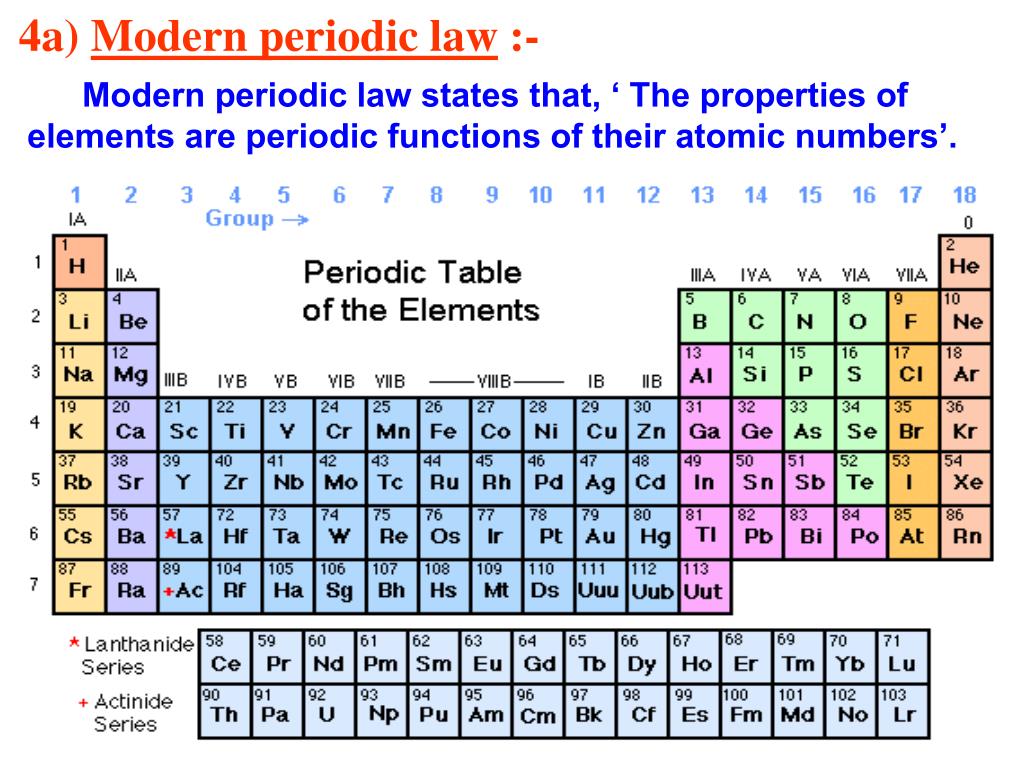
The unreactive nature of the group 18 elements is due to their inability to gain or lose electrons due to their full outer shell.“Table J is also used for electrochemical cell interpretation.” Metals at the top are most easily oxidized and Nonmetals at top are most easily reduced. All the gases in group 18 are similar in properties being heavy, unreactive and having full outer shells. This is in contrast the elements in Group 18 (Noble Gases) are relatively unreactive and are all gases. It is these similarities in reactivity that have allowed scientists to group chemicals together into groups based on their chemical and physical similarities. Lithium will react with water giving a small fizz and bubbling but when reacting caesium with water it reacts violently exploding and creating a lot of heat. this can be seen in the observations of the reaction. All group 1 metals will react with water to produce a hydroxide solution and hydrogen gas but progressing down the group 1 the reaction gets more violent as the outer electron (valence electron) is lost a lot more easily. There are 18 groups in the periodic table spanning from left to right.Īn example of this is the reaction of Group 1 metals with water. Scientists constructing groups of the periodic table were able to study how elements reacted with other elements and spot common similarities and differences. When referring to reactivity of an element this is its relative ease of creating a compound when reacting with another element to form a compound, this often relates to how easy that element will lose or gain electrons. The groups are the vertical columns of the periodic table. The modern periodic table is not only constructed on the increasing atomic number but also using the work of Newlands, elements are divided into groups of similar physical properties and reactivities.
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
